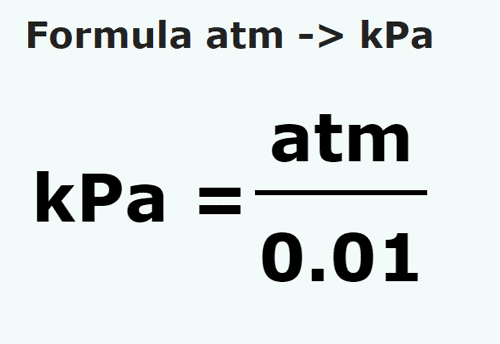SOLVED: What is the pressure of a gas sample when measured to be 654 mmHg? The pressure of the gas sample is 87.2 kPa. The pressure is also given as 6.63 *

Gas Pressure Unit Conversions - torr to atm, psi to atm, atm to mm Hg, kpa to mm Hg, psi to torr - YouTube

VLE data from (i) the UNIQUAC predictive model at 101.3 kPa (1 atm) and... | Download Scientific Diagram

CHEMISTRY (Picture attached) Complete the pressure unit conversions in the table. The first row is - brainly.com

Gas Laws Chapter 5. Pressure Force per unit area Measured in Atmospheres ( atm) Mm of Hg = Torr Pascals or kiloPascals (Pa or kPa) - ppt download

SOLVED: Convert pressure measurements from one unit to another. Given that: 1 atm = 760 mmHg = 14.7 psi = 101.325 kPa 320 mmHg into atm 30.0 atm into kPa
Solved: 1atm=1.01325bar=101.325kPa=760mmHg=760 torr =14.696 Convert the following pressures into t [algebra]